Genetics of CLCN4
The genetics of CLCN4, that is how the condition is caused, and how it can be passed on in families, is complicated.

Are males and females affected differently?
Males
Males only have one X chromosome: they inherit an X chromosome from their mother and a Y chromosome from their father. This means males only have one copy of the CLCN4 gene – they have no ‘back up’ copy if they have a glitch in the CLCN4 gene.
Males with a CLCN4 gene change (either inherited from their mother or due to a de novo change) will have symptoms of CLCN4 – but these symptoms can range widely in severity and frequency.
For females it is more complicated!
Females have two X chromosomes: they inherit one X chromosome from their mother, and another X chromosome from their father. This means that they potentially have a ‘back up’ working copy of the CLCN4 gene. However, in general females only have one functional copy of an X chromosome in each cell due to a process called ‘X chromosome inactivation’. Our current understanding is that how severely a female will be affected by CLCN4 depends on what proportion of the cells in her brain inactivate the copy of the X chromosome with the CLCN4 glitch, and what proportion of the cells in her brain inactivate the copy of the X chromosome with the working copy of the gene.
How severely a female will be affected seems to be also influenced by whether the gene change happened for the first time when they were conceived (a de novo change: see below) or whether the gene change was inherited.
Females who inherit an X chromosome with a CLCN4 gene change are often called ‘carriers’. Female carriers may have no symptoms at all, or may have some symptoms of CLCN4-related condition, usually (but not always) on the mild end of the spectrum. Some female carriers may, for example, have mild learning or speech problems or an increased chance of mental health conditions (anxiety and/ or depression).
On the other hand, females with a de novo CLCN4 gene change often have a similar range of symptoms to males.
So in summary:
Males with a CLCN4 glitch always have symptoms, females with a CLCN4 glitch could be completely unaffected, have mild symptoms or more severe symptoms. They are more likely to be affected if the gene change happened de novo (when they were conceived) rather than being inherited from a parent. We still have a lot to learn to understand this complexity better.
How can children get a CLCN4 related condition?
Children can either have a de novo or an inherited CLCN4-related condition. To know for sure which is the situation in your family, gene testing of parents is needed. After parental testing this information should be included in your child’s gene test report.
De Novo CLCN4 genetic changes.
An individual may be born with a new (de novo) genetic change in the CLCN4 gene, which is not inherited from their mother or father. This is usually because the CLCN4 glitch happened for the first time when the baby was conceived (when the egg and sperm came together to form a baby). CLCN4 related condition was not caused by anything that a parent did or did not do, such as taking certain medications or environmental exposures, it just happens by chance. It was no one’s ‘fault’.
There is a very low chance (estimated at 1 in 100) that the siblings of a child with a de novo CLCN4 glitch will also be affected by CLCN4. However, the chance is not zero, as very occasionally the glitch occurred in a patch of eggs in the mother or sperm in the father (germline mosaicism).
Males with a de novo CLCN4 genetic change will experience symptoms of CLCN4 related condition. Females with a de novo CLCN4 genetic change seem to show a similar range of symptoms to males.
Inherited CLCN4 genetic changes
Alternatively, an individual may inherit a CLCN4 genetic change on an X chromosome from a parent. This means a parent is either affected by, or a carrier of CLCN4. About ¾ of the time, males inherit their CLCN4 gene change from their mother.
What are the chances of a female carrier having a child with a CLCN4 related condition?
People with CLCN4 or who are a carrier of CLCN4 have choices when planning children and these options can be discussed further with a clinical geneticist.
When a female carrier is having children, for each pregnancy, there is a 50% chance of passing on the X chromosome with the working CLCN4 gene and a 50% chance of passing on the X chromosome with the CLCN4 gene change (see diagram below).
This means, for each pregnancy a female carrier of CLCN4 would have:
- 1 in 4 (25%) chance of an affected boy.
- 1 in 4 (25%) chance of a female who is a carrier (may be completely unaffected or mildly affected).
- 1 in 2 (50%) chance of a child who is not affected by CLCN4 and can not pass on CLCN4-related condition to their children.

What are the chances of a male carrier having a child with a CLCN4 related condition?
Males pass on their X chromosome to all their daughters and a Y chromosome to their sons. A male with CLCN4 would, if they have children:
- Pass on the X chromosome with the CLCN4 gene change to all their daughters. This means all their daughters will be carriers and could have children of their own with a CLCN4-related condition.
- Pass on their Y chromosome to all his sons. Therefore none of their sons would be have a CLCN4-related condition, and none of his sons could pass on the condition to their children.
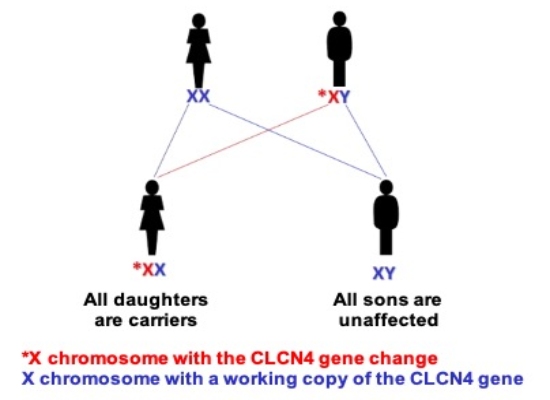
We are aware of at least one male with CLCN4 who has fathered daughters, and those daughters have mild learning difficulties.
How do you test for a CLCN4 gene change?
Genetic testing can be arranged by a Paediatrician or a Clinical Geneticist. DNA is extracted from a blood or saliva sample and tested for a genetic change in the CLCNC4 gene. There are now a range of different types of genetic tests that could be used, and your Paediatrician or Geneticist can advise. It would be important to include a test that not only looks for ‘spelling changes’ (missense, stop-gain, frameshift and splicing changes) in the gene but also small and large chromosomal deletions. Examples could be a chromosomal micoarray and an exome, or whole genome sequencing. More information about genetic testing can be found here.

